Advertisements
Advertisements
प्रश्न
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
उत्तर
The C – O – H bond angle in alcohols is slightly less than the tetrahedral angle (109.5°) because of larger repulsions between the lone pairs of electrons. For example in methanol C – O – H bond angle is 108.9°.
In ethers, the C – O – C bond angle is slightly greater than tetrahedral angle. For example in dimethyl ether C – O – C bond angle is 111.7°. The larger bond angle in ethers may be because of greater repulsions between bulkier alkyl groups as compared to one H in alcohols.
APPEARS IN
संबंधित प्रश्न
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
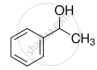
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
The correct geometry around oxygen in CH3OCH3 is
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
