Advertisements
Advertisements
प्रश्न
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
उत्तर
In tert-butyl halides, elimination is favoured over substitution, so alkene is the only reaction product and ether is not formed.
\[\begin{array}{cc}
\phantom{....}\ce{CH3}\phantom{.........}\ce{CH3}\phantom{.......................................}\ce{CH3}\phantom{}\\
\phantom{.}|\phantom{.............}|\phantom{..........................................}|\phantom{}\\
\ce{CH3-C-Br + \overset{+}{N}a\overset{-}{O}C-CH3 -> CH3-C=CH2 + NaBr + CH3-C-OH}\\
\phantom{.}|\phantom{.............}|\phantom{..................}|\phantom{........................}|\phantom{}\\
\phantom{}\ce{\underset{(tert-butyl bromide)}{CH3}}\phantom{....}\ce{CH3}\phantom{..........}\ce{\underset{(2-methylprop-1-ene)}{CH3}}\phantom{..............}\ce{CH3}\phantom{...}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
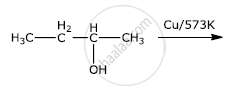
Write the mechanism of the following reaction :
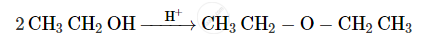
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
By which of the following methods alcohol can be prepared in excellent yield?
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 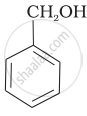 |
| (b) | 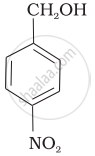 |
| (c) | 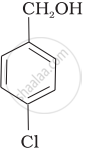 |
Name the factors responsible for the solubility of alcohols in water.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
