Advertisements
Advertisements
प्रश्न
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
उत्तर
Pyridinium chlorochromate (PCC), C5H5NH+Cr CrO3Cl− (in CH2Cl2) or pyridinium dichromate (PDC), \[\ce{(C5H5N^+H)2Cr2O^-_7}\] (in CH2Cl2) is used in the oxidation of a primary alcohol to aldehyde.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
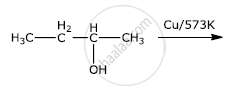
Write the mechanism of the following reaction :
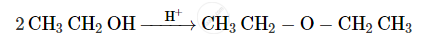
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{.......................}\ce{Br}\\
\phantom{......................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
\phantom{.}|\phantom{......}|\phantom{......................}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{...}\ce{OH}\phantom{...................}\ce{CH3}\phantom{.....}
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
Lucas reagent is ____________.
Lucas test is used for the detection of _____________.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which of the following is not true in case of reaction with heated copper at 300°C?
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
The compound which gives the most stable carbonium ion on dehydration is:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 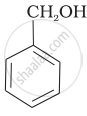 |
| (b) | 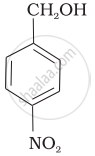 |
| (c) | 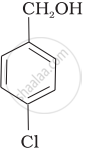 |
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
