Advertisements
Advertisements
प्रश्न
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
उत्तर
Pyridinium chlorochromate (PCC), C5H5NH+Cr CrO3Cl− (in CH2Cl2) or pyridinium dichromate (PDC), \[\ce{(C5H5N^+H)2Cr2O^-_7}\] (in CH2Cl2) is used in the oxidation of a primary alcohol to aldehyde.
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
The compound which reacts fastest with Lucas reagent at room temperature is:
Which of the following is not true in case of reaction with heated copper at 300°C?
Primary and secondary alcohols on the action of reduced copper give:
The compound which gives the most stable carbonium ion on dehydration is:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
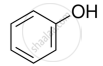
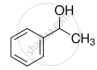
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 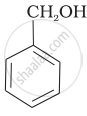 |
| (b) | 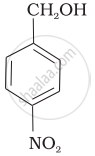 |
| (c) | 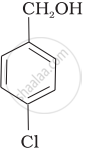 |
What is Lucas reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
