Advertisements
Advertisements
प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Explain why is ortho nitrophenol more acidic than ortho methoxyphenol?
उत्तर
Due to the strong –R and –I effect of the NO2 group, the electron density on the O-H bond decreases; hence, the proton is released easily.
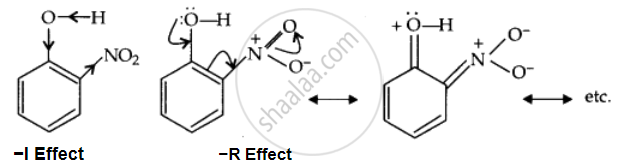
After losing the proton, the remaining o-nitrophenoxide ion becomes stable by resonance.
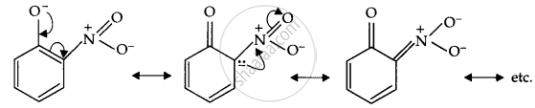
Ortho-nitrophenoxide ion resonance is stable; hence, o-nitrophenol is a strong acid. On the other hand, due to the +R effect of the OCH3 group, the electron density on the O-H bond increases; hence, the removal of the proton becomes difficult.
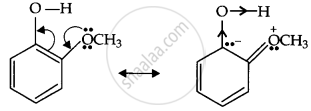
Now, the o-methoxyphenoxide ion, which remains after the loss of the proton, gets destabilized due to resonance.
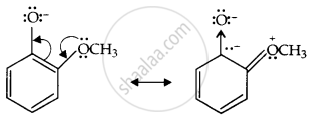
The two negative charges repel each other and destabilize the o-methoxyphenoxide ion, so o-nitrophenol is more acidic than o-methoxyphenol.
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
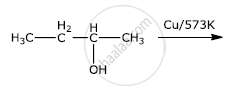
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
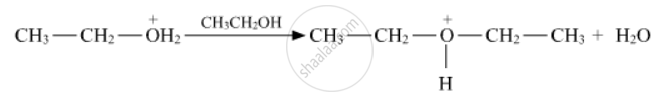
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
Write the mechanism (using curved arrow notation) of the following reaction :
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following is not true in case of reaction with heated copper at 300°C?
Lucas test is done to differentiate between ____________.
Cyclohexene is best prepared from cyclohexanol by which of the following:
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
