Advertisements
Advertisements
प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Explain why is ortho nitrophenol more acidic than ortho methoxyphenol?
उत्तर
Due to the strong –R and –I effect of the NO2 group, the electron density on the O-H bond decreases; hence, the proton is released easily.
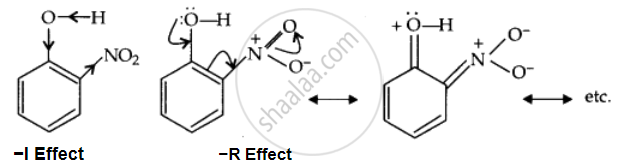
After losing the proton, the remaining o-nitrophenoxide ion becomes stable by resonance.
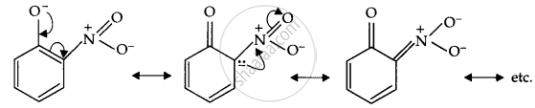
Ortho-nitrophenoxide ion resonance is stable; hence, o-nitrophenol is a strong acid. On the other hand, due to the +R effect of the OCH3 group, the electron density on the O-H bond increases; hence, the removal of the proton becomes difficult.
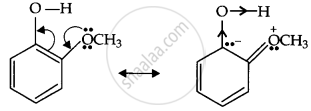
Now, the o-methoxyphenoxide ion, which remains after the loss of the proton, gets destabilized due to resonance.
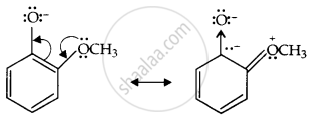
The two negative charges repel each other and destabilize the o-methoxyphenoxide ion, so o-nitrophenol is more acidic than o-methoxyphenol.
संबंधित प्रश्न
Write the mechanism of the following reaction :
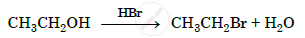
Show how will you synthesize pentan-1-ol using a suitable alkyl halide.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
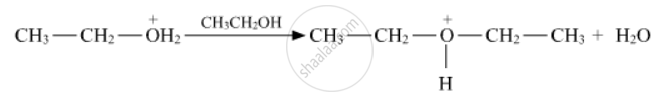
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is used for the detection of _____________.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Dehydration of 2-butanol yields:
Which one of the following on oxidation gives a ketone?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
The process of converting alkyl halides into alcohols involves ______.
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
