Advertisements
Advertisements
प्रश्न
Write the mechanism of the following reaction :
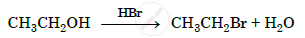
उत्तर १
Primary alcohols such as ethanol react by SN2 mechanism with halogen acid such as HBr as follows:
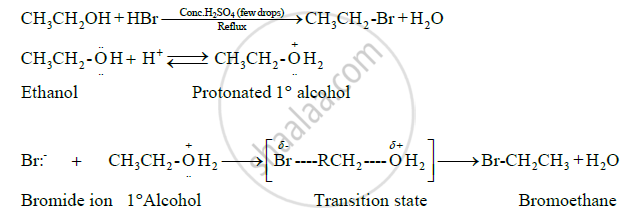
उत्तर २
This is a nucleophilic substitution reaction. It follows SN2 mechanism. The mechanism is as follows:
1) Protonation of hydroxyl group to make it a good leaving group.
2) Displacement of water molecule by bromide ion to form alkyl bromide.
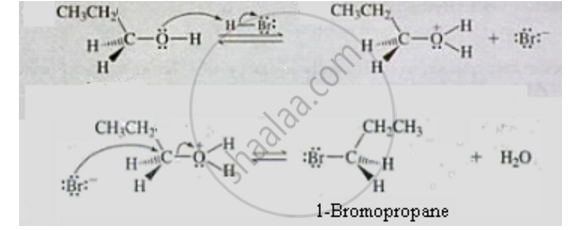
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :
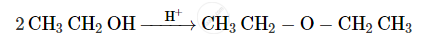
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Which of the following is not true in case of reaction with heated copper at 300°C?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
The process of converting alkyl halides into alcohols involves ______.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
