Advertisements
Advertisements
प्रश्न
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
उत्तर
In phenol, oxygen atom is attached to sp2-hybridised carbon atom while in alcohol, it is attached to sp3-hybridised carbon atom. The bond formed between oxygen and sp2-hybridised carbon is more strongly held then that formed between oxygen and sp3-hybridised carbon.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :
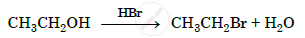
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
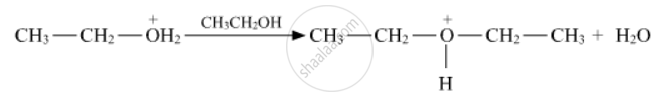
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
The compound which gives the most stable carbonium ion on dehydration is:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Suggest a reagent for the following conversion.

What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
