Advertisements
Advertisements
प्रश्न
Explain why nucleophilic substitution reactions are not very common in phenols.
उत्तर
The C – O bond in phenols has some double-bond character due to resonance and hence cannot be easily cleaved by nucleophiles. So, nucleophilic substitution reactions are not very common in phenols and they give many electrophilic substitution reactions.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
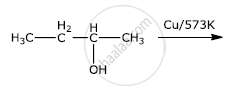
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Lucas test is used for the detection of _____________.
Lucas test is done to differentiate between ____________.
Which one of the following on oxidation gives a ketone?
Primary and secondary alcohols on the action of reduced copper give:
Name the factors responsible for the solubility of alcohols in water.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
