Advertisements
Advertisements
प्रश्न
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
उत्तर
The mechanism of the reaction involves the following three steps:
Step 1: Protonation of alkene to form carbocation by the electrophilic attack of H3O+.
\[\ce{H2O + H+ -> H3O+}\]
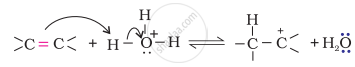
Step 2: Nucleophilic attack of water on carbocation.
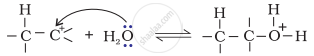
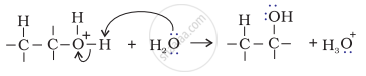
Step 3: Deprotonation to form an alcohol.
APPEARS IN
संबंधित प्रश्न
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Write the structure of 3-Bromo-2-methylprop-1-ene
Name the suitable alcohol and reagent, from which 2-Chloro-2-methyl propane can be prepared.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Short Answer Question.
Identify the product (s) is/are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
