Advertisements
Advertisements
प्रश्न
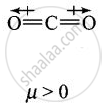
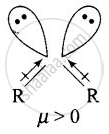
Explain why is \[\ce{O = C =O}\] nonpolar while \[\ce{R - O - R}\] is polar.
उत्तर
\[\ce{O = C =O}\] molecule is linear so that the polarities of two C – O bonds get cancelled and the molecule is linear.
Ethers have structures similar to water and have angular or bent structure. Therefore, the polarity of two R – O groups does not get cancelled and these have net dipole moment. Thus, \[\ce{R - O - R}\] is polar.
APPEARS IN
संबंधित प्रश्न
\[\ce{CH3 - CH = CH2 ->[H2O/H^+]}\]
Write the structure of the product of the following reaction:
\[\begin{array}{cc}
\ce{CH3 - CH2 - CH - CHO ->[NaBH4]}\\
|\phantom{....}\\
\ce{CH3}\phantom{.}
\end{array}\]
Write the structure of the compound whose IUPAC name is as follows:
2-Methylbutan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
2, 3-Diethylphenol
Write the structure of the compound whose IUPAC name is as follows:
1-Ethoxypropane
\[\ce{CH3-O-CH(CH3)2 + HI ->}\] Products is/are:
What is the structure and IUPAC name of glycerol?
When phenol is treated with bromine water, white precipitate is obtained. Give the structure and the name of the compound formed.
Write the structures of the isomers of alcohols with molecular formula \[\ce{C4H10O}\]. Which of these exhibits optical activity?
Glycerol is used
