Advertisements
Advertisements
प्रश्न
Explain why nucleophilic substitution reactions are not very common in phenols.
उत्तर
The C – O bond in phenols has some double-bond character due to resonance and hence cannot be easily cleaved by nucleophiles. So, nucleophilic substitution reactions are not very common in phenols and they give many electrophilic substitution reactions.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
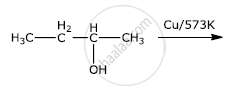
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Dehydration of 2-butanol yields:
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
