Advertisements
Advertisements
प्रश्न
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
उत्तर
\[\ce{\underset{propan-1-ol}{CH3CH2CH2OH} + 2[O] ->[Alkaline KMnO4][Oxidation] \underset{Propanoic acid}{CH3CH2COOH} + H2O}\]
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :
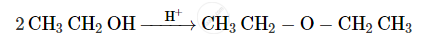
Write the mechanism of the following reaction :
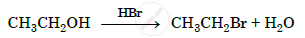
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which one of the following on oxidation gives a ketone?
Primary and secondary alcohols on the action of reduced copper give:
The compound which gives the most stable carbonium ion on dehydration is:
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
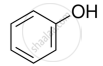
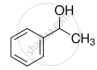
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Explain why nucleophilic substitution reactions are not very common in phenols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
The correct geometry around oxygen in CH3OCH3 is
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
