Advertisements
Advertisements
Question
Explain why nucleophilic substitution reactions are not very common in phenols.
Solution
The C – O bond in phenols has some double-bond character due to resonance and hence cannot be easily cleaved by nucleophiles. So, nucleophilic substitution reactions are not very common in phenols and they give many electrophilic substitution reactions.
APPEARS IN
RELATED QUESTIONS
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
The compound which reacts fastest with Lucas reagent at room temperature is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which of the following is not true in case of reaction with heated copper at 300°C?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 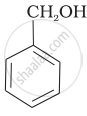 |
| (b) | 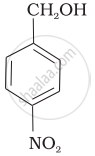 |
| (c) | 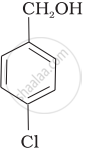 |
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
