Advertisements
Advertisements
प्रश्न
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
विकल्प
catalytic hydrogenation
treatment with \[\ce{LiAlH4}\]
treatment with pyridinium chlorochromate
treatment with \[\ce{KMnO4}\]
उत्तर
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by treatment with pyridinium chlorochromate.
Explanation:
Alcohols are oxidized to aldehydes and finally to acids.
\[\ce{CH3CH2OH ->[{[O]}] CH3CHO ->[{[O]}] CH3COOH}\]
Oxidations can be stooped at aldehyde stage by using pyridinium chlorochromate
\[\ce{(CrO3C5H5N.HCl)}\].
\[\ce{CH3CH2OH ->[PCC] CH3CHO}\]
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :
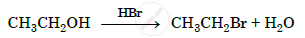
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
Dehydration of 2-butanol yields:
Which of the following is not true in case of reaction with heated copper at 300°C?
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
