Advertisements
Advertisements
Question
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Options
catalytic hydrogenation
treatment with \[\ce{LiAlH4}\]
treatment with pyridinium chlorochromate
treatment with \[\ce{KMnO4}\]
Solution
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by treatment with pyridinium chlorochromate.
Explanation:
Alcohols are oxidized to aldehydes and finally to acids.
\[\ce{CH3CH2OH ->[{[O]}] CH3CHO ->[{[O]}] CH3COOH}\]
Oxidations can be stooped at aldehyde stage by using pyridinium chlorochromate
\[\ce{(CrO3C5H5N.HCl)}\].
\[\ce{CH3CH2OH ->[PCC] CH3CHO}\]
APPEARS IN
RELATED QUESTIONS
Write the mechanism of the following reaction :
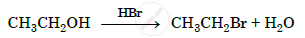
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Dehydration of 2-butanol yields:
Lucas test is done to differentiate between ____________.
In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Which of the following observation is shown by 2-phenyl ethanol with Lucas Reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
