Advertisements
Advertisements
Question
Write the mechanism of the following reaction :
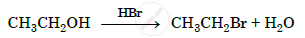
Solution 1
Primary alcohols such as ethanol react by SN2 mechanism with halogen acid such as HBr as follows:
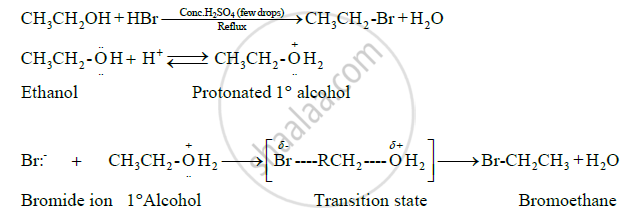
Solution 2
This is a nucleophilic substitution reaction. It follows SN2 mechanism. The mechanism is as follows:
1) Protonation of hydroxyl group to make it a good leaving group.
2) Displacement of water molecule by bromide ion to form alkyl bromide.
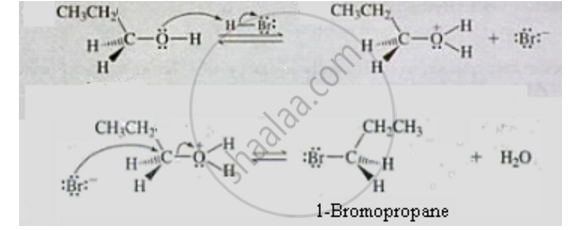
APPEARS IN
RELATED QUESTIONS
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Lucas test is used for the detection of _____________.
The compound which reacts fastest with Lucas reagent at room temperature is:
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Which one of the following on oxidation gives a ketone?
Alcohols react with active metals e.g. Na, K etc. to give corresponding alkoxides. Write down the decreasing order of reactivity of sodium metal towards primary, secondary and tertiary alcohols.
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
