Advertisements
Advertisements
प्रश्न
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
उत्तर
The mechanism of acid dehydration of ethanol to yield ethene involves the following three steps:
Step 1: Protonation of ethanol to form ethyl oxonium ion:
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{.........................}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{..}\\
|\phantom{....}|\phantom{..........................}|\phantom{....}|\phantom{....}|\phantom{..}\\
\ce{H - C - C - \overset{\bullet\bullet}{\underset{\bullet\bullet}{O}} - H + H^+ ⇌[Fast] H - C - C - \underset{\bullet\bullet\phantom{...}}{O^+} - H}\\
\phantom{....}|\phantom{....}|\phantom{..........................}|\phantom{....}|\phantom{..........}\\
\ce{\underset{Ethanol}{H\phantom{...}H}}\phantom{.....................}\ce{\underset{(Ethyl oxonium ion)}{\underset{Protonated alcohol}{H\phantom{...}H}}}\phantom{..}
\end{array}\]
Step 2: Formation of carbocation (rate determining step): It is the slowest step and hence the rate-determining step of the reaction.
\[\begin{array}{cc}
\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{...............}\ce{H}\phantom{....}\ce{H}\phantom{....}\\
|\phantom{....}|\phantom{....}|\phantom{................}|\phantom{.....}|\phantom{....}\\
\ce{H - C - C - \underset{\bullet\bullet\phantom{...}}{O^+} - H <=>[Slow] H - C - C^+ + H2O}\\
|\phantom{....}|\phantom{.....................}|\phantom{....}|\phantom{....}\\
\ce{H}\phantom{...}\ce{H}\phantom{....................}\ce{H}\phantom{...}\ce{H}\phantom{....}
\end{array}\]
Step 3: Elimination of a proton to form ethene:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{....}\ce{H}\phantom{........}\ce{H}\phantom{.}\\
|\phantom{....}|\phantom{......}\backslash\phantom{......}/\\
\ce{H - C - C^+ <=> C = C + H^+}\\
|\phantom{....}|\phantom{......}/\phantom{......}\backslash\\
\phantom{..}\ce{H}\phantom{...}\ce{H}\phantom{....}\ce{\underset{Ethene}{H\phantom{........}H}}\phantom{.}
\end{array}\]
The acid consumed in step 1 is released in Step 3. After the formation of ethene, it is removed to shift the equilibrium in a forward direction.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
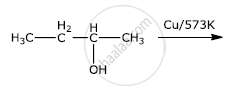
Write the mechanism of the following reaction :
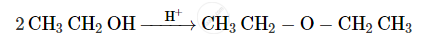
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Which of the following is not true in case of reaction with heated copper at 300°C?
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
The correct geometry around oxygen in CH3OCH3 is
Which of the following alcohols will not undergo oxidation?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
