Advertisements
Advertisements
प्रश्न
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
उत्तर १
If propene is allowed to react with water in the presence of an acid as a catalyst, then propan-2-ol is obtained.
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2}->[H2O/H+][\Delta]CH3 - CH - CH3}\\
\phantom{.........................}|\\
\phantom{............................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
उत्तर २
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2} + Conc.H2SO4 -> CH3 - CH - CH3 ->[H2O, \Delta][-H2SO4] CH3 - CH - CH3}\\
\phantom{.....................................}|\phantom{..........................}|\\
\phantom{..........................................}\ce{OSO3H}\phantom{.................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
Name the reagents used in the following reactions:

Write the main product(s) in each of the following reactions:
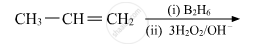
Name the reagents used in the following reactions:
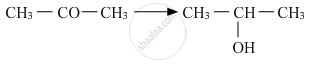
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?
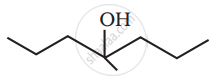
Show how you would synthesise the following alcohol from an appropriate alkene?
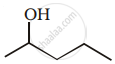
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Which of the following reacts with NaOH to give alcohol?
Identify ‘C’ in the following:
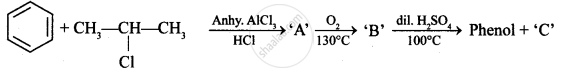
How can propan-2-one be converted into tert- butyl alcohol?
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Explain a process in which a biocatalyst is used in industrial preparation of a compound known to you.
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
Write the mechanism of acid dehydration of ethanol to yield ethene.
