Advertisements
Advertisements
प्रश्न
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
उत्तर १
If propene is allowed to react with water in the presence of an acid as a catalyst, then propan-2-ol is obtained.
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2}->[H2O/H+][\Delta]CH3 - CH - CH3}\\
\phantom{.........................}|\\
\phantom{............................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
उत्तर २
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2} + Conc.H2SO4 -> CH3 - CH - CH3 ->[H2O, \Delta][-H2SO4] CH3 - CH - CH3}\\
\phantom{.....................................}|\phantom{..........................}|\\
\phantom{..........................................}\ce{OSO3H}\phantom{.................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:
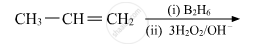
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
Write the mechanism of hydration of ethene to yield ethanol.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
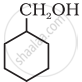
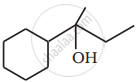
Show how you would synthesise the following alcohol from an appropriate alkene?
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Identify ‘C’ in the following:
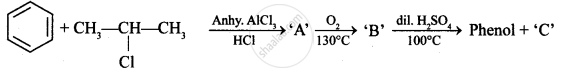
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
When glycol is heated with dicorboxylic acid the product are
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
How are the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
