Advertisements
Advertisements
प्रश्न
Write the mechanism of hydration of ethene to yield ethanol.
उत्तर
The mechanism of hydration of ethene to yield ethanol involves the following three steps:
Step 1: Protonation of alkene to form carbocation by electrophilic attack of H3O+.
\[\ce{H2O + H+ -> H3O+}\]
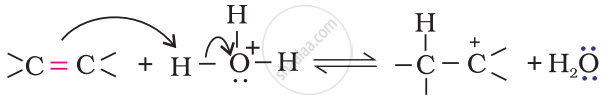
Step 2: Nucleophilic attack of water on carbocation.
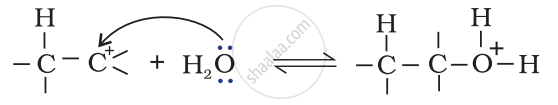
Step 3: Deprotonation to form an alcohol.
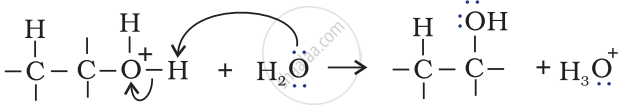
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
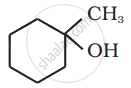
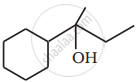
Show how you would synthesise the following alcohol from an appropriate alkene?
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Write the structure of main compounds A and B in the following reaction:
\[\ce{CH3CH2CN->[CH3MgBRH/3O+]A->[LiAIH4]B}\]
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Which of the following reacts with NaOH to give alcohol?
Identify ‘C’ in the following:
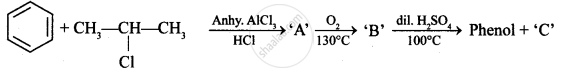
Glycerol as a trimester present in
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
