Advertisements
Advertisements
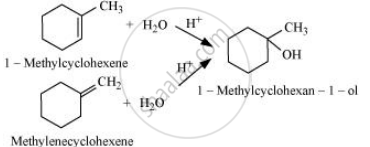
प्रश्न
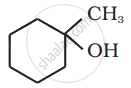
उत्तर
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes.
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
Name the reagent used in the following reaction:
Butan-2-one to butan-2-ol.
Name the reagents used in the following reactions:

Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Monochlorination of toluene in sunlight followed by hydrolysis by aq. \[\ce{NaOH}\] yields.
Benzaldehyde differs from acetaldehyde in that:
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Which of the following reacts with NaOH to give alcohol?
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Glycerol is used in the manufacture
When glycol is heated with dicorboxylic acid the product are
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
How are the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
