Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
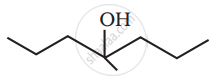
उत्तर
The given alcohols can be synthesized by applying Markovnikov’s rule of acid-catalyzed hydration of appropriate alkenes.
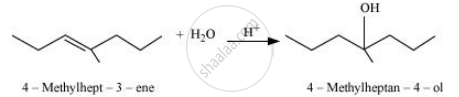
APPEARS IN
संबंधित प्रश्न
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
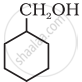
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?
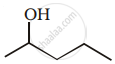
Name the reagents used in the following reactions:

Write the structures of A, B and C in the following reactions :
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
Which of the following reacts with NaOH to give alcohol?
Identify ‘C’ in the following:
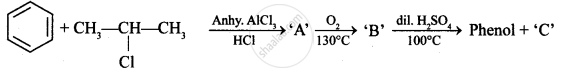
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Most readily hydrolysed halide is:-
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
For distinction between \[\ce{CH3CHO}\] and \[\ce{C6H5CHO}\] the reagent used is ______.
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
Write the mechanism of acid dehydration of ethanol to yield ethene.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide->2-Methylpropan-2-ol}\]
