Advertisements
Advertisements
प्रश्न
You are given benzene, conc. H2SO4 and NaOH. Write the equations for the preparation of phenol using these reagents.
उत्तर १
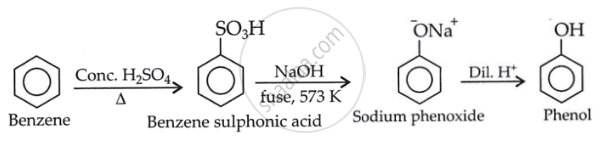
उत्तर २
\[\ce{\underset{Benzene}{C6H6} ->[conc.H2SO4/ \Delta][sulfonation] \underset{Benzene sulphonic acid}{C6H5SO3H} ->[NaOH, melting][-Na2SO3] \underset{Sodium phenoxide}{C6H5ONa} ->[Dil. HCl][-NaCl] \underset{Phenol}{C6H5OH}}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
संबंधित प्रश्न
How do you convert the following:
Aniline to phenol
Write chemical reaction for the preparation of phenol from chlorobenzene.
Write the structures of main products when benzene diazonium chloride (C6 H5 N+2CI−)C6 H5 N2+CI- reacts with the : HBF4/∆
Write the structures of main products when benzene diazonium chloride (C6 H5 N+2CI−)C6 H5 N2+CI- reacts with the :Cu/HBr
What happens when Phenol is oxidised with Na2Cr2O7/H+?
Why phenol undergoes electrophilic substitution more easily than benzene?
Sodium salt of benzene sulphonic acid on fusion with caustic soda gives ____________.
Which of the following will not form phenol or phenoxide?
Name the starting material used in the industrial preparation of phenol.
What happens when reactions:
N-ethylethanamine reacts with benzenesulphonyl chloride.
Benzoic acid reacts with cone. HNO3 and HiSO4 to give:
Aspirin is also known as
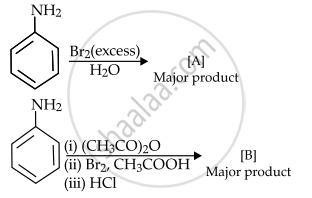
Consider the above reactions, the product A and product B respectively are:
'A' and 'B' in the following reactions are:

A compound 'X' is acidic and it is soluble in NaOH solution, but insoluble in NaHC0 3 solution. Compound 'X' also gives violet colour with neutral FeCI3 solution. The compound 'X' is:
Match List - I with List - II.
| List I | List II | ||
| (A) | 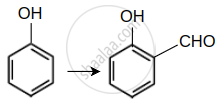 |
(I) | Br2 in CS2 |
| (B) | (II) | Na2Cr2O7/H2SO4 | |
| (C) | 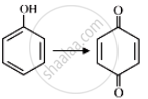 |
(III) | Zn |
| (D) |  |
(IV) | CHCl3/NaOH |
Choose the correct answer from the options given below:
