Advertisements
Advertisements
प्रश्न
Write the mechanism of hydration of ethene to yield ethanol.
उत्तर
The mechanism of hydration of ethene to yield ethanol involves the following three steps:
Step 1: Protonation of alkene to form carbocation by electrophilic attack of H3O+.
\[\ce{H2O + H+ -> H3O+}\]
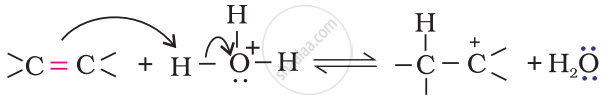
Step 2: Nucleophilic attack of water on carbocation.
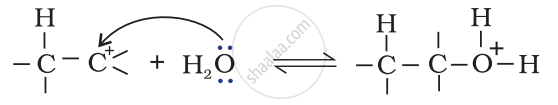
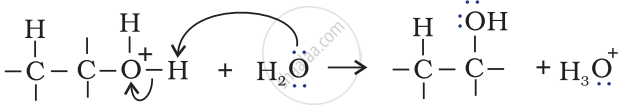
Step 3: Deprotonation to form an alcohol.
APPEARS IN
संबंधित प्रश्न
Write the main product(s) in each of the following reactions:
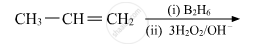
Name the reagents used in the following reactions:
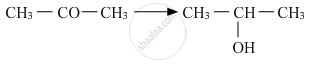
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
What is meant by hydroboration-oxidation reaction? Illustrate it with an example.
Name the reagent used in the following reaction:
Dehydration of propan-2-ol to propene.
Name the reagents used in the following reactions:

Benzaldehyde differs from acetaldehyde in that:
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Alkenes convert into alcohols by ____________.
How can propan-2-one be converted into tert- butyl alcohol?
Select the acid(s) which cannot be prepared by Grignard reagent.
Carboxylic acids are more acidic than phenol and alcohol because of
A compound does not react with 2, 4 dinitrophenylhydrazine, the compound is:
When alcohol react with concentrated H2SO4 intermediate compound formed is
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
Ceric aminonium nitrate and CHCl3/alc. KOH are used for the identification of functional groups present in ______ and ______ respectively.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
