Advertisements
Advertisements
प्रश्न
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
उत्तर
\[\begin{array}{cc}
\phantom{..........}\ce{OMgBr}\\
\phantom{....}|\\
\ce{CH3 - CH - MgBr + HCHO -> CH3 - CH - CH2 ->[H2O] CH3 - CH - CH2OH + Mg(OH)Br}\\
|\phantom{.................................}|\phantom{......................}|\phantom{...................}\\
\phantom{.}\ce{CH3}\phantom{.............................}\ce{CH3}\phantom{...................}\ce{CH3}\phantom{.................}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Name the reagents used in the following reactions:
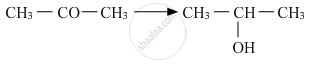
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
Write the mechanism of hydration of ethene to yield ethanol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
Show how you would synthesise the following alcohol from an appropriate alkene?
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Name the reagents used in the following reactions:

Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Alkenes convert into alcohols by ____________.
Which of the following reacts with NaOH to give alcohol?
Commercially carboxylic acids are reduced to alcohols by converting them to the ______.
The Wittig reaction is a reaction between a carbonyl compound (aldehyde or ketone only) and a species known as a phosphoniumylide. What is the expected final product in the Wittig reaction?
Magnesium wire continues to burn in the atmosphere of CO2 because
Alkaline hydrolysis of an alkyl halide can be preferably carried out using ______.
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
The major product of the following reaction is:
\[\begin{array}{cc}
\ce{Cl}\phantom{.........................}\\
|\phantom{..........................}\\
\ce{CH3 - CH - CH3 ->[(i) Alc. KOH][(ii) HBr/peroxide (iii) aq. KOH]}
\end{array}\]
