Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
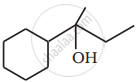
उत्तर
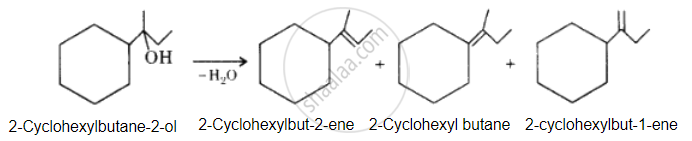
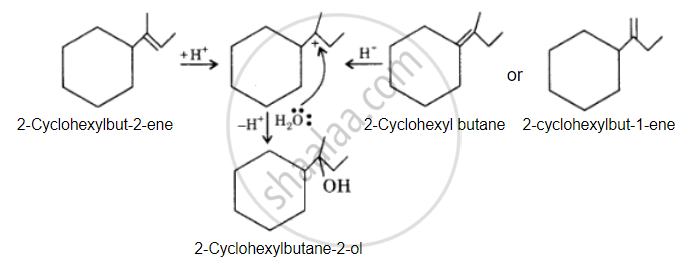
The desired alcohol is obtained from any of these alkenes by adding H2O in the presence of acid.
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:

Predict the major product of acid catalysed dehydration of butan-1-ol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\]
Name the reagents used in the following reactions:

Write the structures of A, B and C in the following reactions :
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Which of the following reacts with NaOH to give alcohol?
Identify ‘C’ in the following:
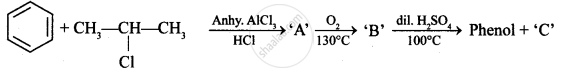
How can propan-2-one be converted into tert- butyl alcohol?
Glycerol as a trimester present in
The best reagent to convert pent - 3 - en - 2 - ol into pent - 3 - en - 2 one is ______.
The major product of acid catalysed dehydration of 1-methylcyclohexanol is ______.
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
