Advertisements
Advertisements
Question
How is the following conversion carried out?
\[\ce{Propene -> Propan-2-ol}\]
Solution 1
If propene is allowed to react with water in the presence of an acid as a catalyst, then propan-2-ol is obtained.
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2}->[H2O/H+][\Delta]CH3 - CH - CH3}\\
\phantom{.........................}|\\
\phantom{............................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
Solution 2
\[\begin{array}{cc}
\ce{\underset{Propene}{CH3 - CH = CH2} + Conc.H2SO4 -> CH3 - CH - CH3 ->[H2O, \Delta][-H2SO4] CH3 - CH - CH3}\\
\phantom{.....................................}|\phantom{..........................}|\\
\phantom{..........................................}\ce{OSO3H}\phantom{.................}\ce{\underset{Propan-2-ol}{OH}}\
\end{array}\]
Notes
Students can refer to the provided solutions based on their preferred marks.
APPEARS IN
RELATED QUESTIONS
How will you convert: Propene to Propan-2-ol?
Write the main product(s) in each of the following reactions:
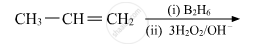
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagents :
CH3MgBr and then H3O+
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Predict the major product of acid catalysed dehydration of butan-1-ol.
Write the mechanism of hydration of ethene to yield ethanol.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
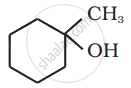
Aldehydes react with Grignard reagent to produce ____________.
Primary alcohols are prepared by the reduction of carboxylic acids. Though lithium aluminium hydride is a strong reducing agent, it is not used in the reaction. This is so because:
Aldehydes are reduced to the corresponding alcohols by the addition of hydrogen in the presence of catalysts to form ____________.
Benzaldehyde differs from acetaldehyde in that:
Alkenes convert into alcohols by ____________.
The reagent used for dehydration of an alcohol is
The best reagent to convert pent 3 – en 2 – 01 into pent 3 – in – 2 – one is
Glycerol is used in the manufacture
To synthesise 1.0 mole of 2-methylpropan-2-ol from Ethylethanoate ______ equivalents of CH3MgBr reagent will be required. (Integer value)
\[\ce{C3H8O ->[{[O]}][K2Cr2O7/H2SO4] C3H6O ->[I2 + NaOH(aq.)] CHI3}\]
In this reaction the first compound is:
\[\ce{? ->[\Delta, CN-][EtOH, H2O]}\] Benzoin.
The reactant is obtained by dry distillation of the calcium salts of the following pairs:
Given below are two statements:
Statement I: On heating with KHSO4, glycerol is dehydrated and acrolein is formed.
Statement II: Acrolein has a fruity odour and can be used to test glycerol's presence.
Choose the correct option.
