Advertisements
Advertisements
प्रश्न
Show how you would synthesise the following alcohol from an appropriate alkene?
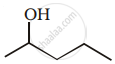
उत्तर
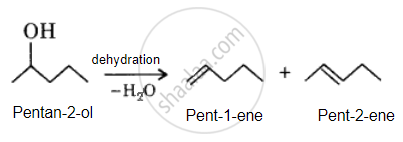
By adding H2O to pent-1-ene, the desired alcohol is obtained.
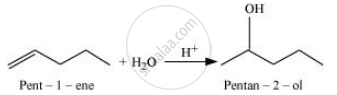
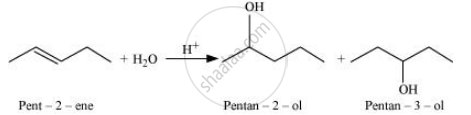
Hence, the desired alkene will be pent-1-ene instead of pent-2-ene.
APPEARS IN
संबंधित प्रश्न
How will you convert: Propene to Propan-2-ol?
Name the reagents used in the following reactions:
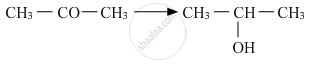
Show how is the following alcohol prepared by the reaction of a suitable Grignard reagent on methanal?
\[\begin{array}{cc}
\ce{CH3 - CH - CH2OH}\\
|\phantom{......}\\
\ce{CH3}\phantom{...}
\end{array}\]
Predict the major product of acid catalysed dehydration of 1-methylcyclohexanol.
Predict the major product of acid catalysed dehydration of butan-1-ol.
Show how will you synthesize 1-phenylethanol from a suitable alkene.
How is the following conversion carried out?
\[\ce{Benzyl chloride -> Benzyl alcohol}\]
How is the following conversion carried out?
\[\ce{Ethyl magnesium chloride -> Propan-1-ol}\]
How will you convert: Phenol to 2, 4, 6 − trinitrophenol?
Ethyl alcohol can be prepared from Grignard reagent by the reaction of ____________.
Why is the reactivity of all the three classes of alcohols with conc. \[\ce{HCl}\] and \[\ce{ZnCl2}\] (Lucas reagent) different?
Carboxylic acids are more acidic than phenol and alcohol because of
Most readily hydrolysed halide is:-
When alcohol react with concentrated H2SO4 intermediate compound formed is

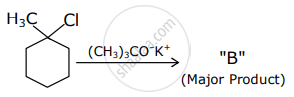
The products "A" and "B" formed in above reactions are:
An aldehyde isomeric with allyl alcohol gives phenyl hydrazone. Pick out a ketone that too gives a phenyl hydrazone containing the same percentage of nitrogen.
How are the following conversion carried out?
\[\ce{Methyl magnesium bromide -> 2-Methylpropan-2-ol}\]
