Advertisements
Advertisements
प्रश्न
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
उत्तर
In tert-butyl halides, elimination is favoured over substitution, so alkene is the only reaction product and ether is not formed.
\[\begin{array}{cc}
\phantom{....}\ce{CH3}\phantom{.........}\ce{CH3}\phantom{.......................................}\ce{CH3}\phantom{}\\
\phantom{.}|\phantom{.............}|\phantom{..........................................}|\phantom{}\\
\ce{CH3-C-Br + \overset{+}{N}a\overset{-}{O}C-CH3 -> CH3-C=CH2 + NaBr + CH3-C-OH}\\
\phantom{.}|\phantom{.............}|\phantom{..................}|\phantom{........................}|\phantom{}\\
\phantom{}\ce{\underset{(tert-butyl bromide)}{CH3}}\phantom{....}\ce{CH3}\phantom{..........}\ce{\underset{(2-methylprop-1-ene)}{CH3}}\phantom{..............}\ce{CH3}\phantom{...}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :
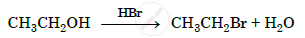
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Lucas reagent is ____________.
\[\ce{CH3CH2OH}\] can be converted into \[\ce{CH3CHO}\] by ______.
Identify the secondary alcohols from the following set:
- \[\ce{CH3CH2CH(OH)CH3}\]
- \[\ce{(C2H5)3COH}\]
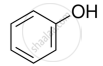
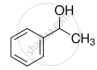
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
What is Lucas reagent?
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
