Advertisements
Advertisements
Question
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Solution
In tert-butyl halides, elimination is favoured over substitution, so alkene is the only reaction product and ether is not formed.
\[\begin{array}{cc}
\phantom{....}\ce{CH3}\phantom{.........}\ce{CH3}\phantom{.......................................}\ce{CH3}\phantom{}\\
\phantom{.}|\phantom{.............}|\phantom{..........................................}|\phantom{}\\
\ce{CH3-C-Br + \overset{+}{N}a\overset{-}{O}C-CH3 -> CH3-C=CH2 + NaBr + CH3-C-OH}\\
\phantom{.}|\phantom{.............}|\phantom{..................}|\phantom{........................}|\phantom{}\\
\phantom{}\ce{\underset{(tert-butyl bromide)}{CH3}}\phantom{....}\ce{CH3}\phantom{..........}\ce{\underset{(2-methylprop-1-ene)}{CH3}}\phantom{..............}\ce{CH3}\phantom{...}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Write the final product(s) in each of the following reactions:
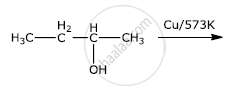
Write the mechanism of the following reaction :
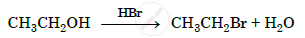
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
The compound which reacts fastest with Lucas reagent at room temperature is:
During dehydration of alcohols to alkenes by heating with cone. H2SO4 the initial step is ____________.
Explain why is OH group in phenols more strongly held as compared to OH group in alcohols.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
