Advertisements
Advertisements
प्रश्न
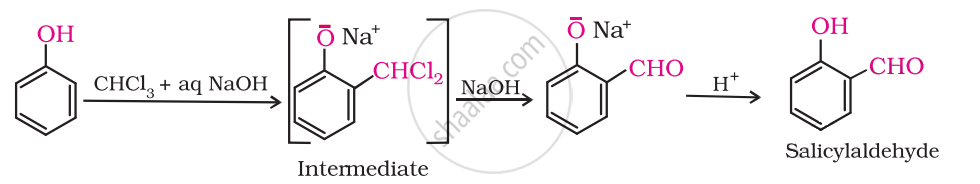
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
उत्तर
APPEARS IN
संबंधित प्रश्न
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Name the reagent used in the following reaction:
Benzyl alcohol to benzoic acid.
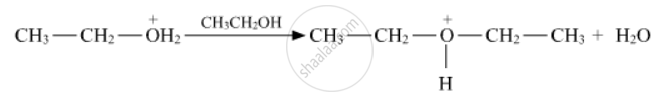
Write the mechanism (using curved arrow notation) of the following reaction :
Lucas test is used for the detection of _____________.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Which of the following are used to convert RCHO into RCH2OH?
(i) H2/Pd
(ii) LiAlH4
(iii) NaBH4
(iv) Reaction with RMgX followed by hydrolysis
Lucas test is done to differentiate between ____________.
Cyclohexene is best prepared from cyclohexanol by which of the following:
Name the factors responsible for the solubility of alcohols in water.
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
What is Lucas reagent?
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
