Advertisements
Advertisements
प्रश्न
Name the factors responsible for the solubility of alcohols in water.
उत्तर
Alcohols are soluble in water because of hydrogen bonding with water molecules.
\[\begin{array}{cc}
\phantom{}\ce{R - O - H ⋅⋅⋅ O - H ⋅⋅⋅ O - H ⋅⋅⋅ }\phantom{}\\
\phantom{.......}|\phantom{..........}|\phantom{.}\\
\phantom{.......}\ce{H}\phantom{.........}\ce{R}\phantom{.}
\end{array}\]
The solubility decreases with increase in size of alkyl or aryl group.
APPEARS IN
संबंधित प्रश्न
Write the final product(s) in each of the following reactions:
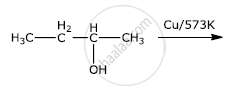
Give reasons for the following:
o-nitrophenol is more acidic than o-methoxyphenol.
Write the mechanism of the following reaction :
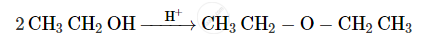
Give the equation of the following reaction:
Oxidation of propan-1-ol with alkaline KMnO4 solution.
Dehydration of 2-butanol yields:
Which of the following is not true in case of reaction with heated copper at 300°C?
Explain why nucleophilic substitution reactions are not very common in phenols.
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
