Advertisements
Advertisements
Question
Name the factors responsible for the solubility of alcohols in water.
Solution
Alcohols are soluble in water because of hydrogen bonding with water molecules.
\[\begin{array}{cc}
\phantom{}\ce{R - O - H ⋅⋅⋅ O - H ⋅⋅⋅ O - H ⋅⋅⋅ }\phantom{}\\
\phantom{.......}|\phantom{..........}|\phantom{.}\\
\phantom{.......}\ce{H}\phantom{.........}\ce{R}\phantom{.}
\end{array}\]
The solubility decreases with increase in size of alkyl or aryl group.
APPEARS IN
RELATED QUESTIONS
Write the mechanism of the following reaction:
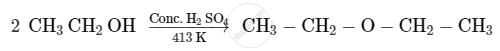
Write the mechanism of the following reaction :
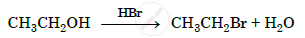
Ortho and para nitrophenols are more acidic than phenol. Draw the resonance structures of the corresponding phenoxide ions.
Give the equation of the following reaction:
Dilute HNO3 with phenol.
Cyclohexene is best prepared from cyclohexanol by which of the following:
Ethers can be prepared by Williamson synthesis in which an alkyl halide is reacted with sodium alkoxide. Di-tert-butyl ether can’t be prepared by this method. Explain.
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
