Advertisements
Advertisements
प्रश्न
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 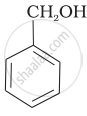 |
| (b) | 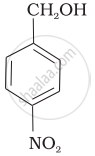 |
| (c) | 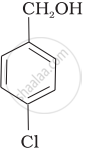 |
विकल्प
a < b < c
b < a < c
b < c < a
c < b < a
उत्तर
b < c < a
Explanation:
It is type of nucleophilic substitution reaction followed by SN1 mechanism. SN1 mechanism depends on the stability of carbonation. Presence of electron-withdrawing group will decrease the stability of carbocation.
APPEARS IN
संबंधित प्रश्न
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to carboxylic acid.
Lucas test is used for the detection of _____________.
Dehydration of 2-butanol yields:
Suggest a reagent for the following conversion.

In Kolbe’s reaction, instead of phenol, phenoxide ion is treated with carbon dioxide. Why?
Assertion: Bond angle in ethers is slightly less than the tetrahedral angle.
Reason: There is a repulsion between the two bulky (–R) groups.
What happens when (CH3)3 C – OH is heated with Cu/573 K?
Write the chemical equation in support of your answer.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene
