Advertisements
Advertisements
प्रश्न
Mark the correct increasing order of reactivity of the following compounds with HBr/HCl.
| (a) | 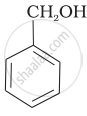 |
| (b) | 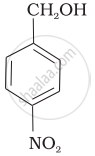 |
| (c) | 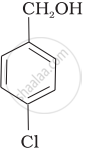 |
पर्याय
a < b < c
b < a < c
b < c < a
c < b < a
उत्तर
b < c < a
Explanation:
It is type of nucleophilic substitution reaction followed by SN1 mechanism. SN1 mechanism depends on the stability of carbonation. Presence of electron-withdrawing group will decrease the stability of carbocation.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :
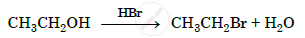
Give the equation of the following reaction:
Treating phenol with chloroform in the presence of aqueous NaOH.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Why is the C – O – H bond angle in alcohols slightly less than the tetrahedral angle whereas the C – O – C bond angle in ether is slightly greater?
The correct geometry around oxygen in CH3OCH3 is
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid-catalysed dehydration of ethanol to yield ethene.
