Advertisements
Advertisements
प्रश्न
Mark the correct order of decreasing acid strength of the following compounds.
| (a) | 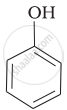 |
| (b) | 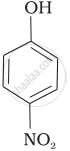 |
| (c) | 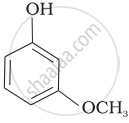 |
| (d) | 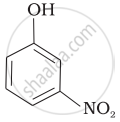 |
| (e) | 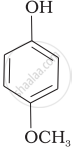 |
पर्याय
e > d > b > a > c
b > d > a > c > e
d > e > c > b > a
e > d > c > b > a
उत्तर
b > d > a > c > e
Explanation:
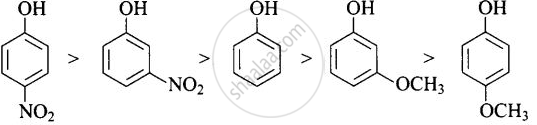
-NO2 is an electron-withdrawing group that increases the acidity of phenol and the effect is more pronounced at ortho and para positions. Similarly, the methoxy group is an electron releasing group that decreases the acidity of phenol and the effect is more pronounced at ortho and para positions.
APPEARS IN
संबंधित प्रश्न
Give two reactions that show the acidic nature of phenol.
The product obtained from the reaction is:
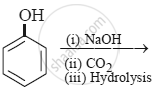
Intermolecular hydrogen bonding is strongest in ______.
Phenols do not react with one of the following:
The ionization constant of phenol is higher than that of ethanol because ____________.
Which of the following statements is true:
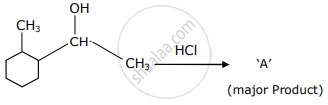
Which is the final product ‘A’ (major) in the given reaction?
For the pair phenol and cyclohexanol, answer the following:
Why is phenol more acidic than cyclohexanol?
Give the structure of the product you would expect when the following alcohol reacts with SOCl2.
2-Methylbutan-2-ol
Compare acidity of phenol with that of ethanol.
