Advertisements
Advertisements
प्रश्न
Which of the following is most acidic?
पर्याय
Benzyl alcohol
Cyclohexanol
Phenol
m-Chlorophenol
उत्तर
m-Chlorophenol
Explanation:
m-chlorophenol is most acidic. Alpha carbon of benzyl alcohol and cyclohexanol is sp3 hybridized. In m-chlorophenol, it is sp2 hybridized. In m-chlorophenol, electron-withdrawing group \[\ce{-Cl}\] is present at meta position.
APPEARS IN
संबंधित प्रश्न
Write IUPAC name of the following compound:
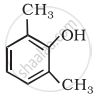
Cresol has ____________.
n-Propyl alcohol and isopropyl alcohol can be chemically distinguished by which reagent?
The product of acid catalysed hydration of 2-phenylpropene is:
Which of the following compounds will react with sodium hydroxide solution in water?
What happens when benzene diazonium chloride is heated with water?
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: Phenol forms 2, 4, 6 – tribromophenol on treatment with \[\ce{Br2}\] in carbon disulphide at 273 K.
Reason: Bromine polarises in carbon disulphide.
Convert the following:
Ethyl alcohol into ethyl acetate
Write chemical reactions for the following conversion:
Acetic acid into ethyl alcohol
