Advertisements
Advertisements
प्रश्न
Which of the following alcohols will not undergo oxidation?
पर्याय
Butanol
Butan-2-ol
2-Methylbutan-2-ol
3-Methylbutan-2-ol
उत्तर
2-Methylbutan-2-ol
Explanation:
Tertiary alcohols will not be under-oxidized. Because 2-Methylbutarte-2-ol is a tertiary alcohol, it will not oxidise.
APPEARS IN
संबंधित प्रश्न
Write the mechanism of the following reaction :
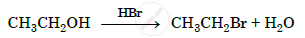
Name the reagent used in the following reaction:
Oxidation of a primary alcohol to aldehyde.
When 3-methylbutan-2-ol is treated with HBr, the following reaction takes place:
\[\begin{array}{cc}
\phantom{.......................}\ce{Br}\\
\phantom{......................}|\\
\ce{CH3 - CH - CH - CH3 ->[HBr] CH3 - C - CH2 - CH3}\\
\phantom{.}|\phantom{......}|\phantom{......................}|\phantom{........}\\
\phantom{}\ce{CH3}\phantom{...}\ce{OH}\phantom{...................}\ce{CH3}\phantom{.....}
\end{array}\]
Give a mechanism for this reaction.
(Hint: The secondary carbocation formed in step II rearranges to a more
stable tertiary carbocation by a hydride ion shift from 3rd carbon atom.)
In the reduction \[\ce{R - CHO + H2 -> RCH2OH}\] the catalyst used is:
Primary and secondary alcohols on the action of reduced copper give:
Cyclohexene is best prepared from cyclohexanol by which of the following:
Suggest a reagent for the following conversion.

Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
Write the mechanism of acid dehydration of ethanol to yield ethene.
