Advertisements
Advertisements
प्रश्न
Explain why low molecular mass alcohols are soluble in water.
उत्तर
The low molecular mass alcohols are soluble in water due to the presence of intermolecular hydrogen bonding between their molecules. Polar O – H group favours dissolution process whereas non-polar alkyl group does not. As the size of the alkyl group increases, it overcomes the effect of the polar nature –OH group and the solubility decreases.
APPEARS IN
संबंधित प्रश्न
Explain the mechanism of the following reaction:
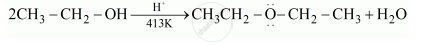
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
The Williamson ether synthesis produces ethers by reacting an
The major product [B] in the following reactions is:
\[\begin{array}{cc}\ce{CH3}\phantom{..................................}\\|\phantom{.....................................}\\\ce{CH3 - CH2 - CH - CH2 - OCH2 - CH3 ->[HI][Heat] [A] alcohol ->[H2SO4][\Delta] [B]}\end{array}\]
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Short Answer Question.
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
Short Answer Question.
Identify the product (s) is/are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
