Advertisements
Advertisements
प्रश्न
Explain why p-nitrophenol is more acidic than phenol.
उत्तर
The electron-withdrawing group (−NO2), withdraws electrons and disperses the negative charge. Therefore, the −NO2 group stabilizes the phenoxide ion. Hence p-nitrophenol is more acidic than phenol.
APPEARS IN
संबंधित प्रश्न
Write the IUPAC name of the given compound:
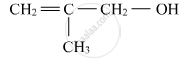
Write structural formulae for 1-Ethylcyclohexanol.
3-methylphenol is called ____________.
IUPAC name of m-cresol is ____________.
Ethylene reacts with Baeyer’s reagent to give ______.
Which of the following is most acidic?
The major product formed by the reaction:
\[\begin{array}{cc}
\ce{CH3CH-CH2Br ->[CH3O^-][CH3OH] is}\\
|\phantom{................}\\
\ce{CH3}\phantom{.............}
\end{array}\]
Give IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - CH2 - CH2 - CH - CH3}\phantom{.}\\
\phantom{.........}|\phantom{...................}|\phantom{...........}\\
\phantom{..}\ce{Cl}\phantom{.................}\ce{OH}\phantom{..}
\end{array}\]
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
