Advertisements
Advertisements
प्रश्न
In one of the industrial processes used for the manufacture of sodium hydroxide, a gas X is formed as by product. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in chemical industry. Identify X and Y giving the chemical equation of the reactions involved.
उत्तर
Sodium hydroxide is manufactured by the electrolysis of a strong solution of sodium chloride (called brine). As a result, chlorine (X) is evolved at anode while hydrogen at cathode. Chlorine reacts with lime water containing slaked lime to form bleaching powder (Y)
`underset(("Lime water"))("Ca"("OH")_2) + underset((X))("Cl"_2) -> underset("Bleaching Powder(Y)")("CaOCl"_2)+"H"_2"O"`
APPEARS IN
संबंधित प्रश्न
State the differences between acids and bases.
When hydrogen chloride gas is prepared on a humid day, the gas is usually passed through the guard tube containing calcium chloride. The role of calcium chloride taken in the guard tube is to:
What is formed when zinc reacts with sodium hydroxide?
How will you protect yourself from the heat generated while diluting a concentrated acid?
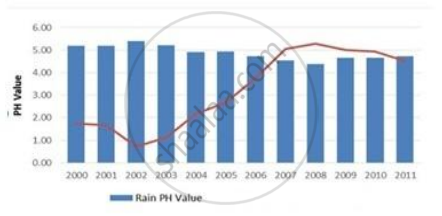
In which year is concentration of hydrogen ion the highest?
Which of the given options correctly represents the parent acid and base of Calcium Carbonate?
How would you distinguish between baking powder and washing soda by heating?
Suggest a safe procedure of diluting a strong concentrated acid.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
Write the chemical equation for the following activity.
Dilute nitric acid was added to calcium oxide.
