Advertisements
Advertisements
प्रश्न
Indicate the steps in the preparation of K2Cr2O7 from chromite ore.
उत्तर
Dichromates are generally prepared from chromate, which in turn is obtained by the fusion of chromite ore \[\ce{(FeCr_2O_4)}\] with sodium or potassium carbonate in free air. The reaction with sodium carbonate occurs as follows:
\[\ce{4FeCr_2O_4 + 8Na2CO3 + 7O_2 ->8Na_2CrO_4 + 2Fe_2O_3 + 8CO2}\]
The yellow solution of sodium chromate is filtered and acidified with sulphuric acid to give a solution from which orange sodium dichromate, \[\ce{Na2Cr2O7. 2H2O}\] can be crystallised.
\[\ce{2Na_2CrO_4 + 2H^+ -> Na_2Cr_2O_7 + 2Na^+ + H_2O}\]
Sodium dichromate is more soluble than potassium dichromate. Therefore, the latter is prepared by treating the solution of sodium dichromate with potassium chloride.
\[\ce{Na_2Cr_2O_7 + 2KCl -> K_2Cr_2O_7 + 2NaCl}\]
Orange crystals of potassium dichromate crystallise out. The chromates and dichromates are interconvertible in an aqueous solution, depending on the pH of the solution. The oxidation state of chromium in chromate and dichromate is the same.
\[\ce{2CrO^{2-}_4 + 2H^+ -> Cr2O^{2-}_7 + H2O}\]
\[\ce{Cr2O^{2-}_7 + 2OH^- -> 2CrO^2-_4 + H2O}\]
Hence, when the pH is increased, making the solution alkaline, dichromate ions (orange in colour) are converted into chromate ions, and the solution becomes yellow.
APPEARS IN
संबंधित प्रश्न
Complete the following equations:
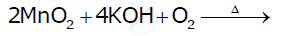
When chromite ore FeCr2O4 is fused with NaOH in presence of air, a yellow-coloured compound (A) is obtained, which on acidification with dilute sulphuric acid gives a compound (B). Compound (B) on reaction with KCl forms an orange coloured crystalline compound (C).
(i) Write the formulae of the compounds (A), (B) and C.
(ii) Write one use of compound (C).
Complete the following equations : MnO4- + 4H+ + 3e- →
Complete the following equation : MnO4- + 8H+ + 5e- →
Name the oxometal anions of the first series of the transition metals in which the metal exhibits the oxidation state equal to its group number.
Give an example and suggest a reason for the following feature of the transition metal chemistry:
A transition metal exhibits the highest oxidation state in oxides and fluorides.
Complete the following equations:
`2MnO_4^(-)+16H^++5S^(2-)rarr`
Answer the following question.
When MnO2 is fused with KOH in the presence of KNO3 as an oxidizing agent, it gives a dark green compound (A). Compound (A) disproportionates in an acidic solution to give a purple compound (B). An alkaline solution of compound (B) oxidizes KI to compound (C) whereas an acidified solution of compound (B) oxidizes KI to (D). Identify (A), (B), (C), and (D).
On addition of small amount of \[\ce{KMnO4}\] to concentrated \[\ce{H2SO4}\], a green oily compound is obtained which is highly explosive in nature. Identify the compound from the following.
When \[\ce{Cu^2+}\] ion is treated with \[\ce{KI}\], a white precipitate is formed. Explain the reaction with the help of chemical equation.
What is the effect of pH on dichromate ion solution?
Potassium dichromate when heated with concentrated sulphuric acid and a soluble chloride, gives brown-red vapours of ______.
Complete the reaction mentioning all the products formed:
\[\ce{2KMnO4 ->[\Delta]}\]
Which of the following ions will have a magnetic moment value of 1.73 BM.
\[\ce{Sc^3+, Ti^3+, Ti^2+, Cu^2+, Zn^2+}\]
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the step in the preparation of K2Cr2O7 from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
Indicate the steps in the preparation of \[\ce{K2Cr2O7}\] from chromite ore.
