Advertisements
Advertisements
प्रश्न
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
K[Cr(H2O)2(C2O4)2]
उत्तर
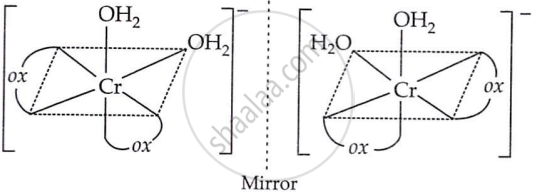
Both geometrical (cis-, trans-) and optical isomers for cis are possible.
(a) Geometrical isomers (cis-trans):
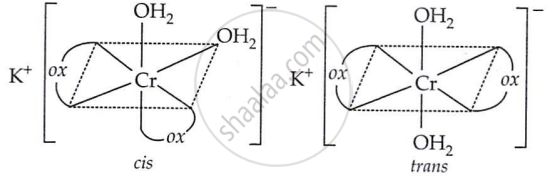
(b) Optical isomers of cis (d- and l-):
APPEARS IN
संबंधित प्रश्न
Explain cationic complexes and anionic complexes of co-ordination compounds.
When a coordination compound CoCl3.6NH3 is mixed with AgNO3, 3moles of AgCl are precipitated per mole of the compound. Write
(i) Structural formula of the complex
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
[Co(en)3]Cl3
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
[Co(NH3)5(NO2)](NO3)2
Draw the structure of optical isomers of [Cr(NH3)2Cl2(en)]+.
Draw all the isomers (geometrical and optical) of [CoCl2(en)2]+.
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
Name the type of isomerism exhibited by the following pairs of compound:
(1) (C2H5)2NH and CH3-NH-C3H7
(2) 1 – butanol and 2 methyl-1 -propanol.
Name the type of isomerism that the compound with molecular formula C3H6O2 exhibits. Represent the isomers.
The complex [(Pt(Py)(NH3)BrCl] will have how many geometrical isomers?
Which one of the following complexes shows optical isomerism? (en = ethylenediamine)
Draw the geometrical isomers of [Co(en)2Cl2]2+. Which geometrical isomer of [Co(en)2Cl2]2+ is not optically active and why?
Which of the following molecules has a chiral centre correctly labelled with an asterisk (*)?
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers:
[Pt(NH3)(H2O)Cl2 ]
Indicate the type of isomerism exhibited by the following complex and draw the structure for this isomer:
[Pt(NH3)(H2O)Cl2]
Indicate the type of isomerism exhibited by the following complex and draw the structure for this isomer: \[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complex and draw the structure for these isomer: \[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the type of isomerism exhibited by the following complex and draw the structure for the isomer:
\[\ce{[Pt(NH3 )(H2O)Cl2]}\]
