Advertisements
Advertisements
प्रश्न
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
[Co(NH3)5(NO2)](NO3)2
उत्तर
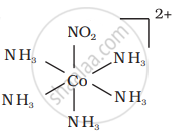
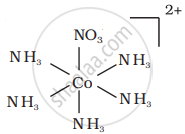
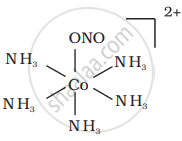
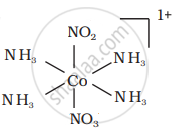
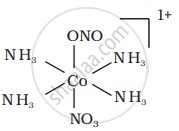
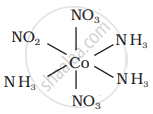
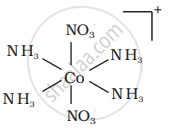
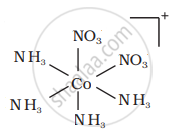
There are 10 possible isomers. (Geometrical, ionisation and linkage isomers)
 |
 |
 |
 |
 |
 |
 |
 |
 |
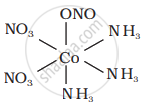 |
It can also show linkage isomerism.
[Co(NH3)5(NO2)](NO3)2 and [Co(NH3)5(ONO)](NO3)2
It can also show ionization isomerism.
[Co(NH3)5(NO2)](NO3)2 and [CO(NH3)5(NO3)](NO3)(NO2)
APPEARS IN
संबंधित प्रश्न
Explain cationic complexes and anionic complexes of co-ordination compounds.
Draw one of the geometrical isomers of the complex [Pt (en)2Cl2] +2 which is optically inactive
Draw the geometrical isomers of complex [Pt(NH3)2Cl2].
What type of isomerism is exhibited by the complex [Co(en)3]3+?
(en = ethane-1,2-diamine)
Define the following term:
Anomers
Indicate the types of isomerism exhibited by the following complex and draw the structures for these isomers:
[Co(en)3]Cl3
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
[Pt(NH3)(H2O)Cl2]
Draw the structure of optical isomers of [Cr(C2O4)3]3−.
Draw the structure of optical isomers of [PtCl2(en)2]2+.
Draw all the isomers (geometrical and optical) of [Co(NH3)2Cl2(en)]+.
Write all the geometrical isomers of [Pt(NH3)(Br)(Cl)(py)] and how many of these will exhibit optical isomers?
What type of structural isomers are [Co(NH3)5 Br] SO4 and [Co(NH3)5 SO4]Br? Give a chemical test to distinguish the isomers.
Write the IUPAC name of [Co(en)2Cl2]+ ion and draw the structures of its geometrical isomers.
Identify the optically active compounds from the following:
(i) \[\ce{[Co(en)3]^{3+}}\]
(ii) \[\ce{[trans - [Co(en)2Cl2]^+}\]
(iii) \[\ce{cis - [Co(en)2Cl2]^+}\]
(iv) \[\ce{[Cr(NH3)5Cl]}\]
The complex [(Pt(Py)(NH3)BrCl] will have how many geometrical isomers?
Indicate the types of isomerism exhibited by the following complexes and draw the structure for isomers:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Indicate the types of isomerism exhibited by the following complexes and draw the structures for these isomers: [Pt(NH3)(H2O)Cl2]
Indicate the types of isomerism exhibited by the following complex and draw the structure for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
