Advertisements
Advertisements
प्रश्न
| It is the phenomenon of the emission of electrons from a metallic surface when light of a suitable frequency is incident on it. The emitted electrons are called photoelectrons. Nearly all metals exhibit this effect with ultraviolet light but alkali metals like lithium, sodium, potassium, cesium, etc. show this effect even with visible light. It is an instantaneous process, i.e., photoelectrons are emitted as soon as the light is incident on the metal surface. The number of photoelectrons emitted per second is directly proportional to the intensity of the incident radiation. The maximum kinetic energy of the photoelectrons emitted from a given metal surface is independent of the intensity of the incident light and depends only on the frequency of the incident light. For a given metal surface, there is a certain minimum value of the frequency of the incident light below which emission of photoelectrons does not occur. |
In a photoelectric experiment, plate current is plotted against anode potential.
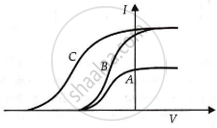
पर्याय
A and B will have the same intensities, while B and C will have different frequencies.
B and C will have different intensities, while A and B will have different frequencies.
A and B will have different intensities, while B and C will have equal frequencies.
B and C will have equal intensities, while A and B will have the same frequencies.
MCQ
घटनेचा अभ्यास
उत्तर
B and C will have equal intensities, while A and B will have the same frequencies.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?
