Advertisements
Advertisements
प्रश्न
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
पर्याय
2 – chloro – 1 – iode – 2 – methyl propane
1 – chloro – 2 – iodo – 2 – methyl propane
1, 2 – dichloro – 2 – methyl propane
1, 2 – diiodo – 2 – methyl propane
उत्तर
2 – chloro – 1 – iode – 2 – methyl propane
APPEARS IN
संबंधित प्रश्न
Read the following reaction and answer the questions given below:
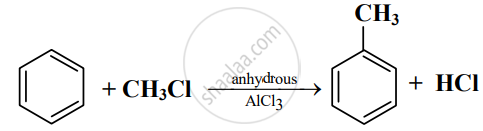
- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Identify the compound (A) in the following reaction
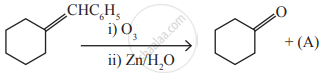
Consider the nitration of benzene using mixed con H2SO4 and HNO3 if a large quantity of KHSO4 is added to the mixture, the rate of nitration will be ______.
Identify the compound A, B, C and D in the following series of reactions.

Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
How will you convert ethyl chloride into n – butane?
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Direct bromination of benzene with excess reagent results in the formation of ____________.
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{.............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
