Advertisements
Advertisements
प्रश्न
Match the structures of the compounds given in Column I with the name of the compounds given in Column II.
| Column I | Column II | |
| (i) | 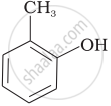 |
(a) Hydroquinone |
| (ii) | 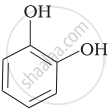 |
(b) Phenetole |
| (iii) | 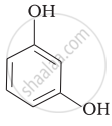 |
(c) Catechol |
| (iv) | 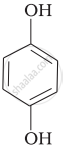 |
(d) o-Cresol |
| (v) | 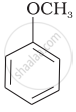 |
(e) guinone |
| (vi) | 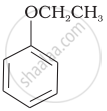 |
(f) Resorcinol |
| (g) Anisole |
उत्तर
| Column I | Column II | |
| (i) |  |
(d) o-Cresol |
| (ii) |  |
(c) Catechol |
| (iii) |  |
(f) Resorcinol |
| (iv) |  |
(a) Hydroquinone |
| (v) |  |
(g) Anisole |
| (vi) |  |
(b) Phenetole |
Explanation:
(i) Cresols are organic compounds which are methyl phenols. There are three forms of cresol: o-cresol, p-cresol and m-cresol.
(ii) Catechol is also known as pyrocatechol. Its IUPAC name is 1, 2-dihydrobenzene. It is used in the production of pesticides, perfumes and pharmaceuticals.
(iii) Its IUPAC name is 1, 3-dihydroxybenzene. Resorcinol is used to treat acne, seborrheic dermatitis and other skin disorder.
(iv) Hydroquinone is also known as quinol. Its IUPAC name is 1, 4-dihydroxybenzene. It is a white granular solid. It is a good reducing agent.
(v) Anisole or methoxy benzene, is a colourless liquid with a smell reminiscent of an anise seed.
(vi) Phenetole is an organic compound. It is also known as ethyl phenyl ether. It is volatile in nature and its vapour is explosive in nature.
APPEARS IN
संबंधित प्रश्न
What is metamerism?
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
Write structural formulae for Methyl vinyl ether.
Write structural formulae for 1-Ethylcyclohexanol.
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
Which of the following gives a positive iodoform test?
\[\ce{Phenol ->[Zn, dust] 'X' ->[CH3Cl][Anhy. AlCl3] 'Y' ->[Alkaline][KMnO4] 'Z'}\]
The product ‘Z’ is:
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Assertion: IUPAC name of the compound
\[\begin{array}{cc}
\ce{CH3 - CH - O - CH2 - CH2 - CH3}\\
|\phantom{....................}\\
\ce{CH3}\phantom{.................}
\end{array}\] is 2-Ethoxy-2-methylethane.
Reason: In IUPAC nomenclature, ether is regarded as hydrocarbon derivative in which a hydrogen atom is replaced by —OR or —OAr group [where R = alkyl group and Ar = aryl group]
Assertion: Like bromination of benzene, bromination of phenol is also carried out in the presence of Lewis acid.
Reason: Lewis acid polarises the bromine molecule.
