Advertisements
Advertisements
प्रश्न
Write IUPAC name of the following compound:
C6H5 – O – C7H15(n−)
उत्तर
1-Phenoxyheptane
APPEARS IN
संबंधित प्रश्न
Name the following compound according to IUPAC system.
\[\begin{array}{cc}
\ce{CH3 - C = C - CH2OH}\\
\phantom{}|\phantom{....}|\phantom{....}\\
\phantom{}\ce{CH3}\phantom{.}\ce{Br}\phantom{...}
\end{array}\]
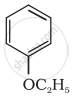
Give IUPAC name of the following ether:
Write the structure and IUPAC name of 'methyl-n-propyl ether'.
How is phenol converted into the following?
benzene
Write IUPAC name of the following compound (CH3)2 N − CH2CH3
Write the structures of the products when Butan-2-ol reacts with SOCl2
Write structural formulae for Methyl vinyl ether.
Write IUPAC names of the following
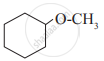
Give IUPAC names of the following compound:
\[\begin{array}{cc}
\phantom{..}\ce{H}\phantom{...}\ce{CH3}\phantom{.}\ce{H}\phantom{..}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\ce{H - C - C - C - H}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{}\\
\phantom{.}\ce{H}\phantom{...}\ce{OH}\phantom{.}\ce{H}\phantom{.}\\
\end{array}\]
IUPAC name of m-cresol is ____________.
IUPAC name of m-cresol is ______.
Which of the following compounds will react with sodium hydroxide solution in water?
Which of the following reagents can be used to oxidise primary alcohols to aldehydes?
(i) \[\ce{CrO3}\] in anhydrous medium.
(ii) \[\ce{KMnO4}\] in acidic medium.
(iii) Pyridinium chlorochromate.
(iv) Heat in the presence of Cu at 573 K.
Write the IUPAC name of the compound given below.
\[\begin{array}{cc}
\phantom{}\ce{CH3 - CH2 - C = C - OH}\\
\phantom{........}|\phantom{....}|\phantom{}\\
\phantom{..............}\ce{CH3 CH2OH}
\end{array}\]
Assertion: Addition reaction of water to but-1-ene in acidic medium yields butan-1-ol.
Reason: Addition of water in acidic medium proceeds through the formation of primary carbocation.
Assertion: p-nitrophenol is more acidic than phenol.
Reason: Nitro group helps in the stabilisation of the phenoxide ion by dispersal of negative charge due to resonance.
Write complete reaction for the bromination of phenol in aqueous and non-aqueous medium.
How are the following conversions carried out?
Methyl magnesium bromide→2-Methylpropan-2-ol.
