Advertisements
Advertisements
प्रश्न
Match the type of packing given in Column I with the items given in Column II.
| Column I | Column II |
| (i) Square close packing in two dimensions |
(a) Triangular voids |
| (ii) Hexagonal close packing in two dimensions |
(b) Pattern of spheres is repeated in every fourth layer |
| (iii) Hexagonal close packing in three dimensions |
(c) Coordination number 4 |
| (iv) Cubic close packing in three dimensions |
(d) Pattern of sphere is repeated in alternate layers |
उत्तर
| Column I | Column II |
| (i) Square close packing in two dimensions |
(c) Coordination number 4 |
| (ii) Hexagonal close packing in two dimensions |
(a) Triangular voids |
| (iii) Hexagonal close packing in three dimensions |
(d) Pattern of sphere is repeated in alternate layers |
| (iv) Cubic close packing in three dimensions |
(b) Pattern of spheres is repeated in every fourth layer |
Explanation:
(i) Square close packing in two dimensions each sphere have coordination number 4, as shown below.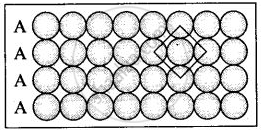
(ii) Hexagonal close packing in two dimensions each sphere has coordination number 6 as shown below and creates a triangular void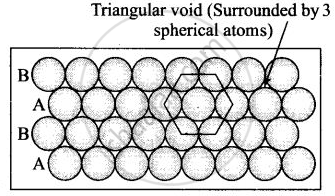
(iii) Hexagonal close packing in 3 dimensions is a repeated pattern of sphere in alternate layers also known as ABAB pattern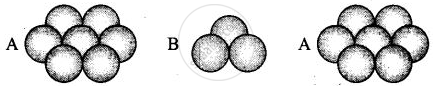
(iv) Cubic close packing in a 3 dimensions is a repeating pattern of sphere in every fourth layer.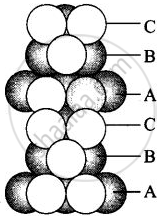
APPEARS IN
संबंधित प्रश्न
A compound forms hexagonal close-packed structure. What is the total number of voids in 0.5 mol of it? How many of these are tetrahedral voids?
The coordination number of AB having rock salt geometry is ____________.
Hexagonal close packed arrangement of ions is described as ____________.
The arrangement ABC ABC.... is referred to as ____________.
Which of the following statement is not true about the hexagonal close packing?
How can you best describe the elongated octahedral structure of blue vitriol, CuSO4.5H2O?
The octane number of Iso-octane is
Co-ordination number of sodium ion Na+ in Nacl is:-
The right options for the number of tetrahedral and octahedral voids in the hexagonal primitive unit cells is ______.
The density of a pure substance 'X' whose atoms pack in cubic close pack arrangement is 1 g/cc. If all tetrahedral voids are occupied by 'Y' atoms. The value of '3a' is ______ g/cc, if the density of resulting solid is 'a' g/cc.
[Given: Atomic mass (X) = 30 g/mol, (Y) = 20 g/mol]
